Affordable, custom-tailored solutions to support your trials.
Companies developing innovative products need innovative CROs. This demand is our credo. Our team of scientists, medical doctors and programmers never considered tradition as synonym for correctness. Our clients get the best of our human skills and the best innovative software tools to run clinical studies more cost effective, more solid and more transparent.
GCP-Service has developed a Quality Controlled Trial Management System (QCTMS), which offers much more than other comparable systems.
QCTMS is an effective risk management tool in accordance with the amended ICH E6 (R2).
QCTMS – is a highly flexible framework that supports all relevant activities of clinical trials, e.g. planning, start-up, monitoring, and administration.
The use of QCTMS not only improves quality effectively, but also saves resources by saving man-power.

Quality Controlled Trial Management System (QCTMS®)
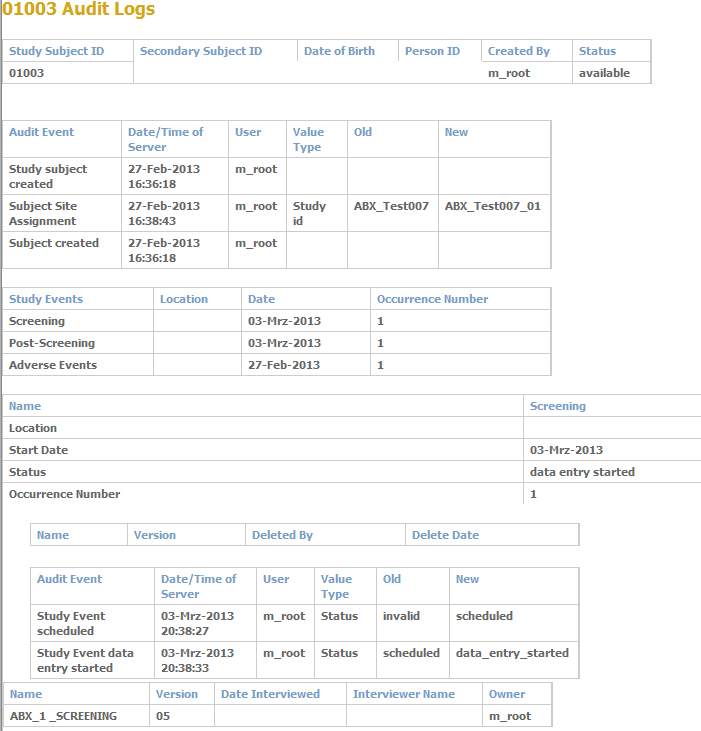
Is a flexible system which can be specifically adapted to each clinical study, sponsor or SOP. Individual tolerance alert level can be specified to initiate preventive actions, or as an audit trigger to initiate corrective and preventive actions at the first sign of their occurrence. Typical parameters, such as informed consent process, SAE reporting timelines, recruitment rates, query rates, number of protocol deviations, monitoring frequency, statistical parameters and many additional factors, can be used to evaluate the risk with regards to data quality, or risks affecting study subjects. Different risk levels trigger automatic alerts to your Project Management and/or Quality Assurance department.
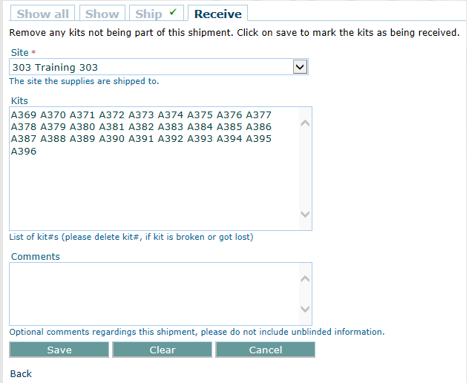
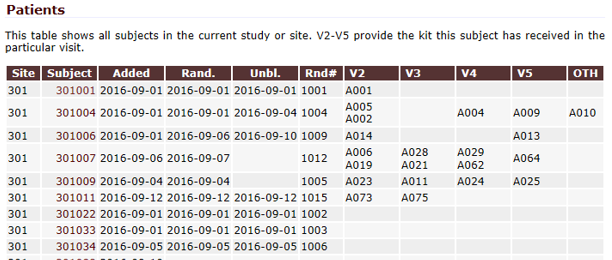
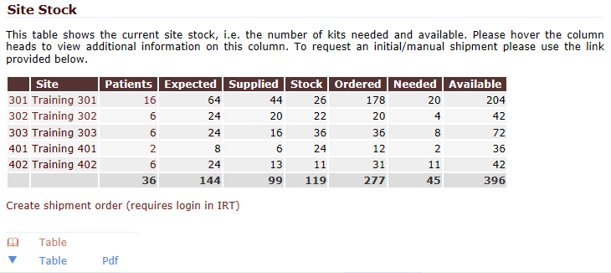
QCTMS® IRT (Interactive Response Technology)
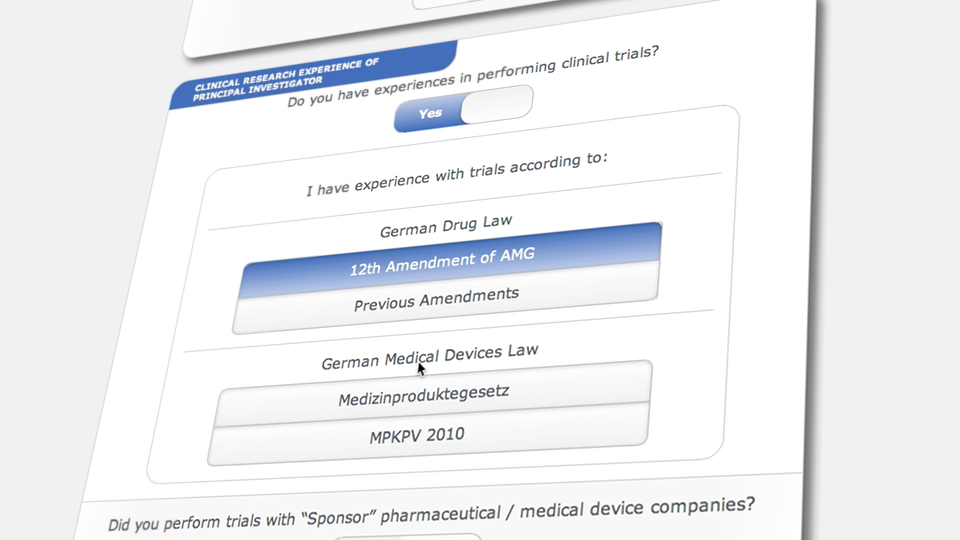
GCP-Service has developed QCTMS IRT to sponsors and CROs to use web based randomization or drug assignment solution in a simple and extremely cost effective way.
The interactive response technology of QCTMS® IRT allows centralized web-based randomization and drug assignment in an extremely user friendly way. We know that site personnel are normally not very comfortable in sophisticated IT environments; this fact alone allows us to develop a simple, easy-to-use electronic tool ensuring compliance by every user.
The advantages of QCTMS IRT are:
• Flexible system which can be used for every study design
• Up to 5 stratification parameters in one trial
• Easy to use, intuitive and user-friendly
• Connection to QCTMS for quality control of randomization data
• Controlled code break option
• 21 CFR Part 11 compliant
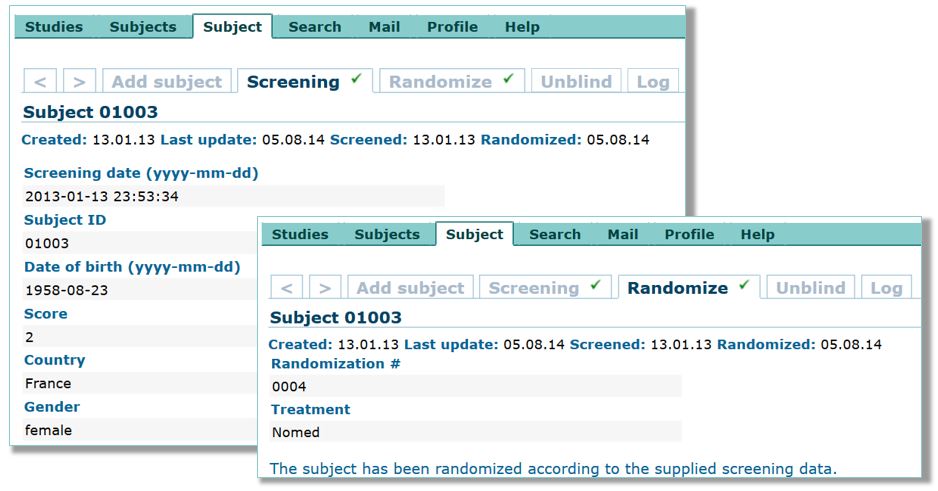
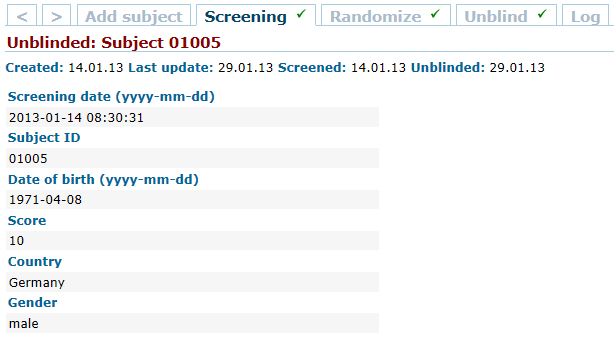
QCTMS® EDC (Electronic Data Capture)
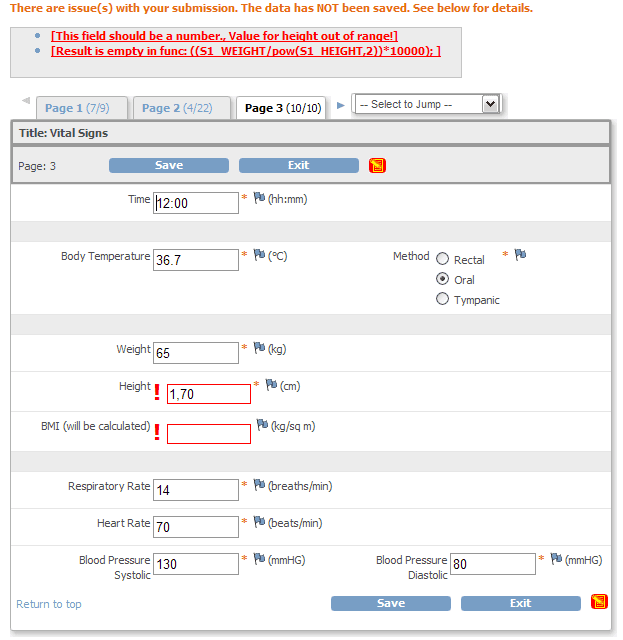
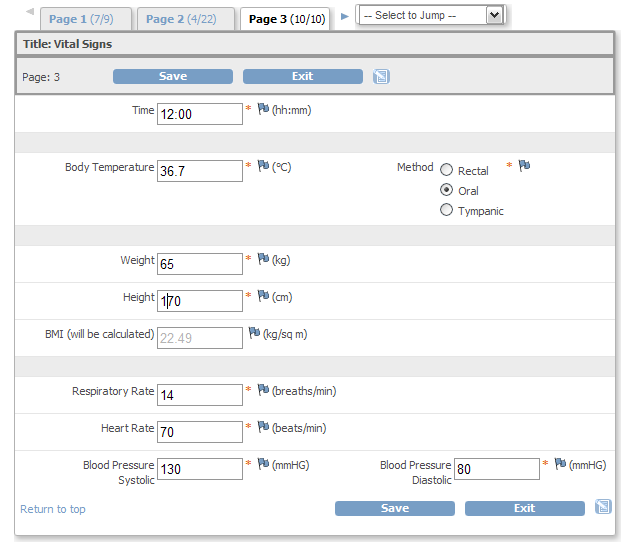
QCTMS® EDC allows physicians, researchers and their personnel to specifically and individually capture the research data to be acquired and to analyze them in a standardized manner. Our EDC system provides the following features and benefits:
• Timely and rapid data acquisition and reporting
• Integrated data check for completeness and plausibility
• Integrated query management
• Standardized documentation of adverse events
• Individually configurable roles and authorizations
• Simple and safe to use thanks to our “next steps” philosophy
• Data output in any desired format
Our EDC system is extremely cost effective which makes it a useful tool even for clinical studies with a small number of patients. The combination of our EDC with our Clinical Trial Management System (QCTMS) improves quality but also saves money by saving man-power. After a very short time, project teams are able to recognise the benefit of these unique systems.
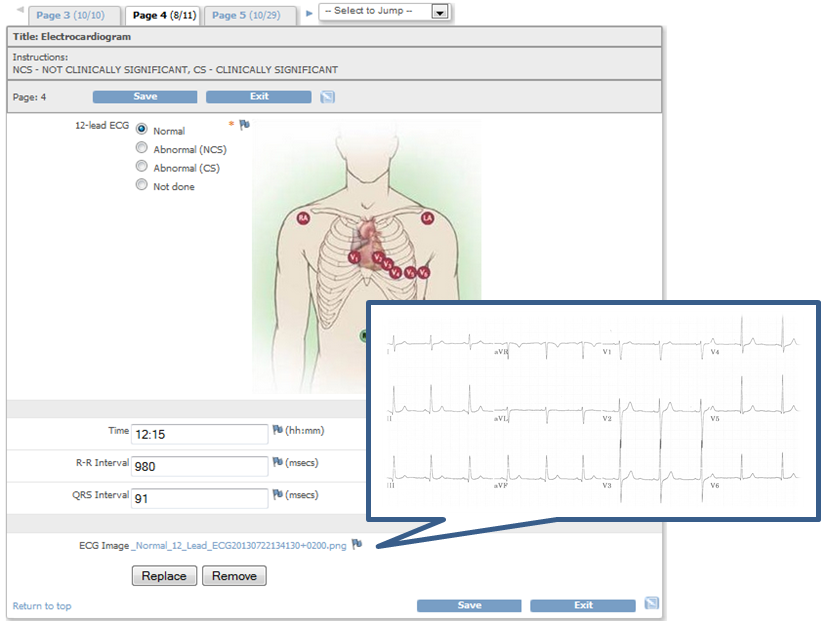
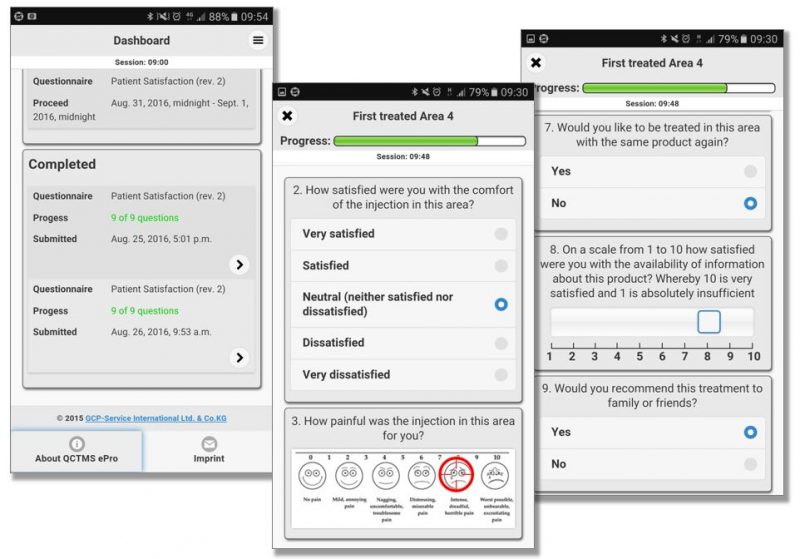
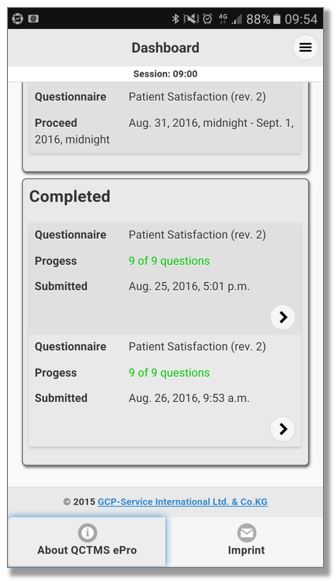
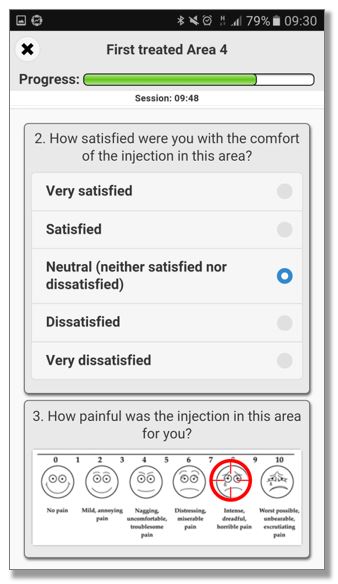
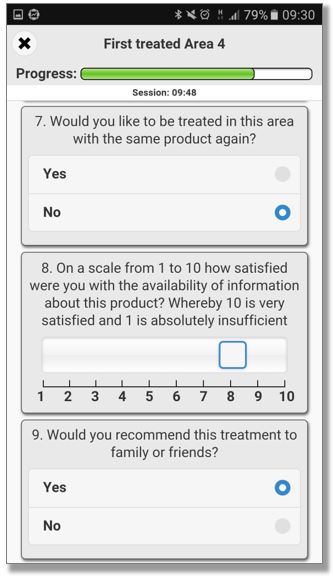
QCTMS® ePRO (electronic Patient Reported Outcome)
This smart and easy-to-use tool allows centralized data entry for patient diaries and questionnaires in a user friendly way. We know that patients in clinical trials are often not familiar with IT environments. Therefore we developed a simple electronic tool ensuring compliance by every user. QCTMS® ePRO allows study subjects to capture the trial data in a fast and error-free way and provides the site staff with the required oversight. Integrated control functions save manual queries and ensure a higher data quality and integrity.
The main features of the QCTMS® ePRO are the following:
• Timely and rapid data acquisition and reporting
• Integrated data check for completeness and plausibility
• Individually configurable roles and authorizations
• Overall data quality improvement
• Available in all languages
• Cost effectiveness
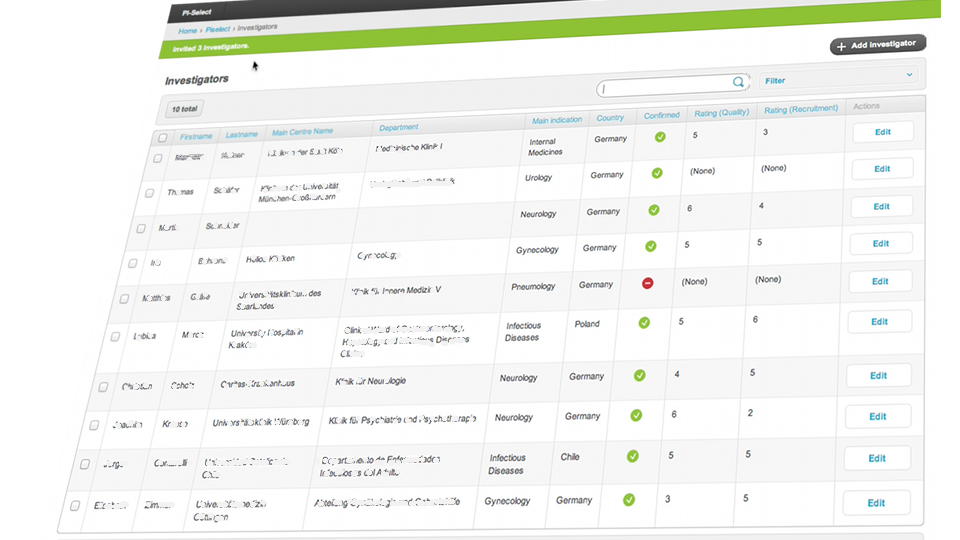
Project Feasibilities
With our support we help our clients to streamline the process of conducting reliable and high quality feasibilities.
Keeping timelines in clinical studies is always critical. Therefore, an effective feasibility and an excellent site selection process are crucial in the early stage of clinical trials. Carrying out high quality feasibility reviews and study site selections for large clinical trials are extremely time and cost intensive. For multicentre studies, around 3-4 hours per study site need to be spent to receive reliable feasibility data. GCP-Service’s PI-Select system is a very effective site selection and feasibility tool that significantly reduces manpower, time and costs for the essential start-up phase by delivering excellent results.