In every clinical trial, statistics play a very important part. Because based on the statistical analysis, decisions are made about new therapies, medicaments, or medical products on a binary level. But of course, the data could also yield a certain percentage showing that an outcome was positive when in fact it should have been negative. But fortunately, these errors can be accounted for before starting with the conduction of the study!
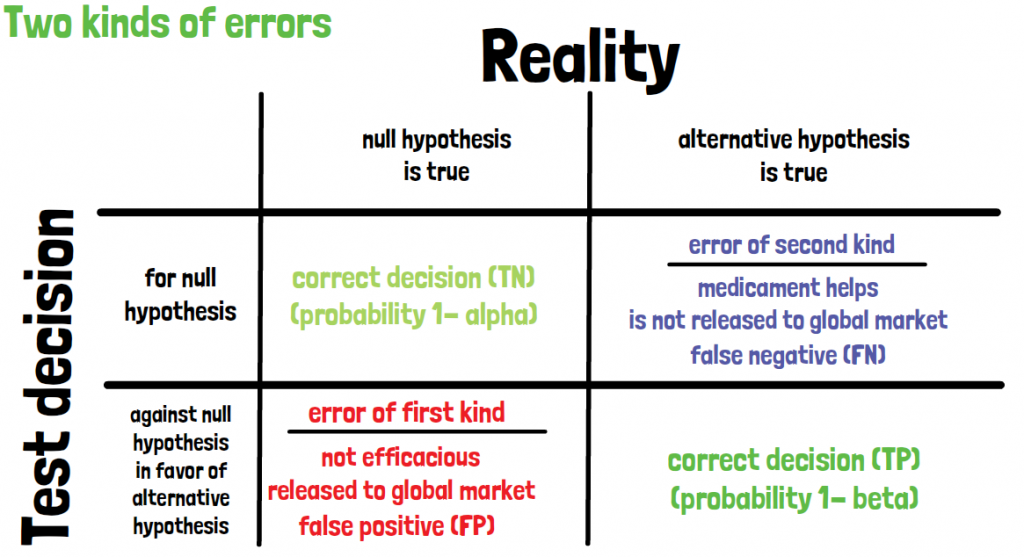
There are two kind of errors that can occur. The first being a medicament that is released to the global market, which is not efficacious. This is also called a false positive outcome, or error of the first kind. The second error being exactly the opposite, a medicament that helps is not released to the market. That is also called false negative or error of the second kind. In a cross table it can be visualized as follows: in the rows the decision of the test conducted is stated, and in the columns the true outcome of the two hypotheses is shown. The error of the first kind is given when the null hypothesis is true, but the test would reject it. The error of the second kind would be if the alternative hypothesis is true but the test is deciding in favor of the null-hypothesis.
When presented graphically, the two treatment groups are distinguished between their expected means but show similar variances. In the overlapping area false positive and false negative results can occur. Important here is to define the critical value, a threshold that distinguishes the two treatment groups. That part of the left treatment group that lies on the right of this critical value is the proportion of falsely accepting the null-hypothesis. That part of the right treatment arm that lies on the left of the critical value represents the proportion of the falsely rejected null hypothesis.
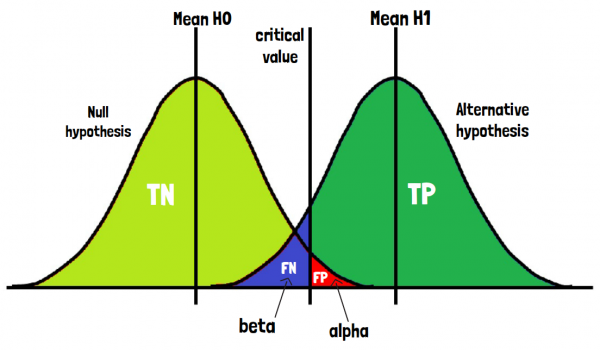
The latter kind of error is the error of the first kind or also called alpha error or type I error. It is one of the crucial factors that biostatisticians include already in the beginning of a clinical trial, for example in the sample size calculation. The probability of falsely accepting the null-hypothesis is also called beta error or type II error. Equally to the alpha error it is determined in the planning phase of a clinical trial when considering the power of the statistical test. The beta error is calculated by subtracting the power from 100%.
If you are interested in a visual presentation of this topic, please feel free to follow us on our GCP Mindset YouTube channel! If you would like to know more about how we could implement statistics in your clinical trial, send us a mail to statistics@gcp-service.com.