It is a widely spread view that biostatisticians should be involved only after data has been collected. But involving a biostatistician already in the planning phase can efficiently save time and money throughout the whole duration of the trial. In the planning phase, a biostatistician has an important impact on the structure and type of the study. The definition of the objective of the trial is done by considering scientific and logistical aspects, to determine which objective is most likely to result in a successful and meaningful trial. Here, the biostatistician advises on endpoints for the trial, which should be objectively measurable and clearly defined. It has to be defined if the trial is of confirmatory nature or solely exploratory, as well as the number of treatment arms and how they are embedded in the trial design. Only then the sample size can be calculated by the biostatistician, which has a large influence on the costs, logistics and duration of the clinical trial.
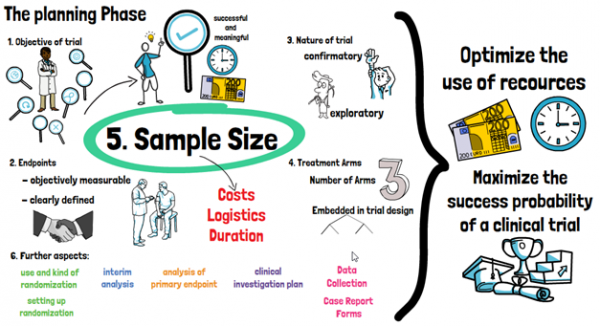
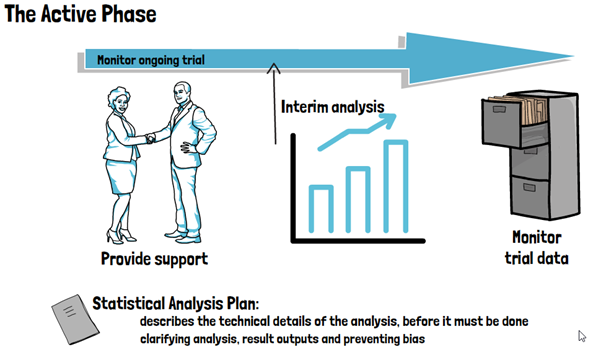
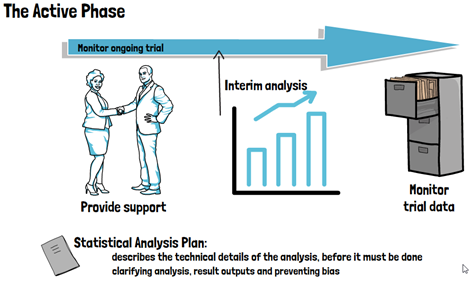
Further aspects in this phase of the trial, that involve biostatisticians, are decisions regarding the use and type of randomization, whether interim analyses can and should be conducted and how exactly the primary endpoint of the trial will be analyzed. All of this will be formalized in a Clinical Study Protocol or Clinical Investigation Plan. As such, involving a biostatistician during the planning phase of the trial can not only optimize the use of resources but also maximize the success probability of a clinical trial. During the active phase of the trial, biostatisticians monitor the ongoing trial and provide support for making decisions that may impact the analysis at the end of the trial. They may further be involved in identifying issues with data quality and integrity, and conducting any interim analyses foreseen for the trial. The biostatistician will also set up a Statistical Analysis Plan, which prospectively describes the technical details of the entire trial analysis in greater (and more technical) detail.
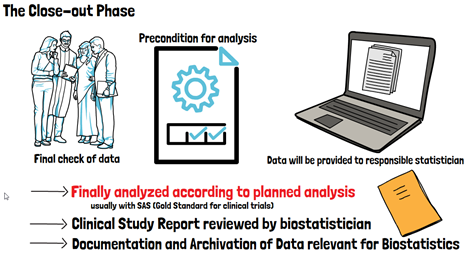
Finally, in the close-out phase a final check of the data and any preconditions for the analysis will usually be performed and the database locked. Only after that, all data will be provided to the biostatistician and analyzed according to the planned analysis. Once the results are available, a Clinical Study Report or Clinical Investigation Report must be written, which is generally also reviewed or co-authored by the biostatistician to confirm that all results were interpreted correctly. At the very end, the biostatistician must also ensure that everything is documented and archived correctly.
If you are interested in a visual presentation of this topic, please feel free to follow us on our GCP Mindset YouTube channel! If you would like to know more about how we could implement statistics in your clinical trial, send us a mail at: statistics@gcp-service.com