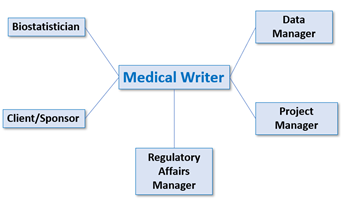
A medical writer is responsible for producing well-structured documents presenting scientific, clinical, educational, or promotional information clearly and concisely. In clinical trials, delivery of these documents requires input from areas such as data management, biostatistics, and regulatory affairs. Data managers supply clinical data to be used by biostatisticians who process them to derive conclusions regarding safety and efficacy of the treatment or device under investigation.
Regulatory affairs managers make sure that a clinical trial is conducted according to regulations and are in charge with communicating with regulatory authorities and ethics committees. Therefore, a medical writer is in constant communication with all these parties to ensure satisfactory generation of clinical trial documents. The interaction with the sponsor/client is also important, especially in the beginning and final stages of document creation. Typically, there are several daily calls with team members and clients to coordinate and clarify different tasks.
Throughout the day, medical writers usually work on several projects in parallel, for example in relation to clinical trials of drugs in different stages of development. They may exist solely as the only writer on a project, or they may be a part of a medical writing team, as in the case of large global phase III trials.
A day usually starts with a review of the work schedule, meetings, and timelines regarding different projects. An indispensable aspect is literature search and review, as medical writers need to be familiar and up- to- date with current research on medical conditions and the evolution of their therapies. This also serves to gather material to be incorporated into introduction and discussion sections of documents such as clinical study protocols and reports.
In addition, local translations and country-specific regulatory adaptations of documents need to be tended to. Close cooperation with biostatisticians is a must, as the interpretation of statistical analysis is crucial for the results and impact of clinical trials.
Usually, not all of the above will happen every single day, but it is nevertheless always a dynamic and gratifying work environment. Lastly, some medical writers also spend a significant portion of their time travelling, for example to congresses where they are involved in producing posters and presentations for expert speakers.


