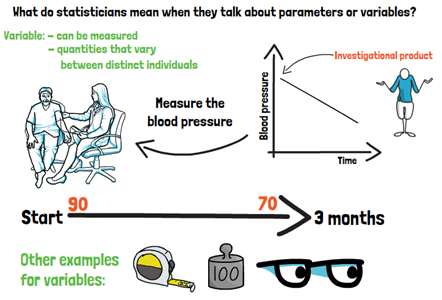
What do statisticians mean when they talk about parameters or variables? In the frame of a clinical study, a variable is something that can be measured – like the blood pressure for example. Let us assume as the main objective of our study, we are interested if the investigational product is decreasing the blood pressure over time. What we can do to answer our question easily, is to measure the blood pressure once in the beginning of the trial and then again, after the patient used the product, after three months. We can compare these values or variables directly. So, variables in general are quantities which vary between distinct individuals, in this case patients.
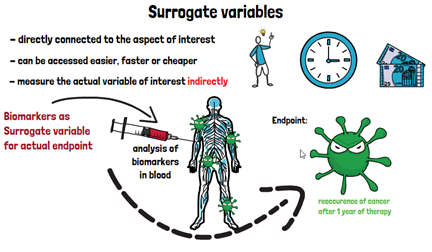
Sometimes it is not possible or too costly to obtain the variable of interest directly. This is when so called surrogate variables come into play. These are variables that are directly connected to the variable of interest but can be assessed easier, faster, or cheaper. Therefore, they are used to measure the actual variable of interest indirectly. In clinical trials, surrogate variables must fulfill certain criteria before they should be used as such: There should be a scientific reason behind assuming that there is a connection between the surrogate and the actual variable of interest, there should be studies of the prognostic value of the surrogate in the general population, and there should be prior clinical trials that have investigated the direct outcome as well as the surrogate. Having only a correlation is not sufficient to serve as a surrogate variable.
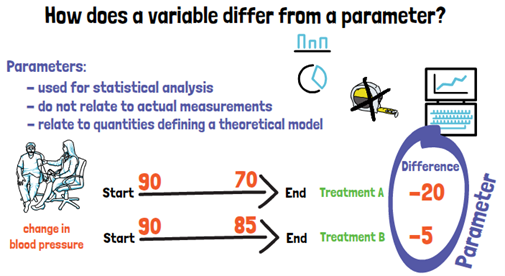
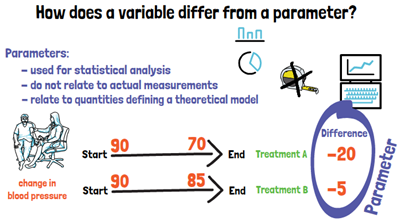
Now that we know what a variable is, how does it differ from a parameter? Parameters are what biostatisticians use for the statistical analysis. They do not relate to actual measurements but to quantities defining a theoretical model. If we look at our example with the blood pressure, here we would calculate the change in blood pressure from the beginning to the end of our trial, for the two treatments. What we compare now between the two treatments are parameters. They cannot be measured directly and were computed from measured variables.
 If you are interested in a visual presentation of this topic, please feel free to follow us on our GCP Mindset YouTube channel! If you have any statistical questions, feel free to contact us at: statistics@gcp-service.com
If you are interested in a visual presentation of this topic, please feel free to follow us on our GCP Mindset YouTube channel! If you have any statistical questions, feel free to contact us at: statistics@gcp-service.com