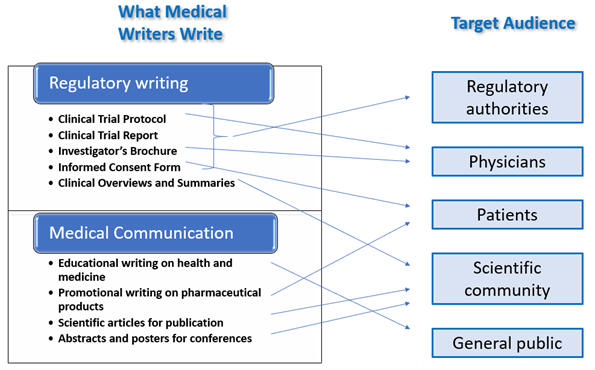
Medical writers communicate clinical data and various findings in medicine to a wide range of audiences and within the scope of a number of different documents. These audiences may include physicians, patients, regulatory authorities, scientific community, and the general public. A medical writer needs to be thoroughly familiar with each of these audiences, considering their level of understanding and their different interests. For example, the wording and format of an Investigator’s Brochure, which aims to explain the background and properties of a drug to a Physician has very different style and requirements from an Informed Consent Form, which must present information to a prospective patient in layman’s terms.
The focus of Regulatory Writing is the development of novel drugs and medical devices with the end goal of obtaining marketing authorization for them. This encompasses documents of formulaic format such as Clinical Trial Protocols, Reports, Investigator’s Brochures and Informed Consent Forms. They are produced at different stages of drug development and need to be approved by bodies such as Ethics Committees and Regulatory Authorities. Besides regulatory and ethics bodies, their designated audience comprises doctors who will administer an experimental treatment, and patients who will receive it.
On the other hand, Medical Communication may provide educational content to the general public on the developments in health and different medical conditions, for example in health magazines. In addition, it may also be more narrowly targeted, and done for promotional purposes focusing on single pharmaceutical companies and their products with the target audience being health care professionals and consumers.
Understanding the duties is just the first step — read about opportunities and career paths in Career as a Medical Writer.