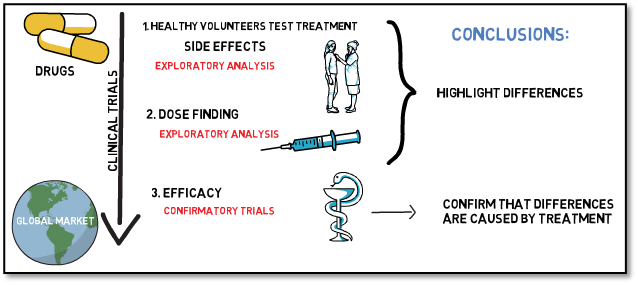
Clinical trials often aim to establish two key parameters about a novel treatment or device: They should show that the product is safe and efficacious. This information culminates in the risk-benefit ratio of a product, which is a deciding factor to determine if a product is released to the market or not. Assuming that, on average, the investigational treatment is better than the comparator: Is this sufficient to say that there is a benefit over the comparator? To answer this question, a statistical test must be performed. It basically answers the question, how likely it is that a difference could have been observed randomly. It is here where we differentiate between exploratory and confirmatory analysis.
The main goal of exploratory analyses is to understand the relation of cause and effect, and therefore allow the assessment of relevant influence factors. Based on the outcome of such analyses, new hypotheses can be developed, and research questions opened. In exploratory analyses we can only describe what we see, but we cannot draw any statistical conclusions on why we see it. That means we can say that we observed a difference between treatment and the comparator, but we cannot say with certainty that this difference is caused by the difference in treatment.
Confirmatory analyses on the other hand evaluate pre-determined relationships, with concrete expectations towards the outcome. Hypotheses must be defined prospectively at the start of the trial and are statistically tested based on pre-defined models or tests. This gives the chance to be able to state that the differences observed are caused by a difference in treatment.
The probability of coincidentally observed differences, where there are none, is increasing with every additional test you perform on the same data. Therefore, generally only one or very few confirmatory analyses are performed for each trial, and additional analyses should be performed in an exploratory manner to not increase this probability.
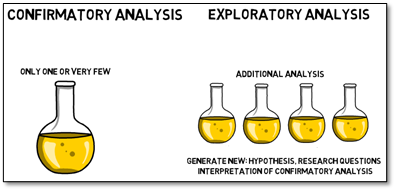
This also means that one must be careful with how to interpret exploratory results. They should only be used to generate new hypotheses and research questions for following trials, and to assist in interpretation of the confirmatory analysis.
If you are interested in a visual presentation of this topic, please feel free to follow us on our GCP Mindset YouTube channel!
If you would like to know more about how we could implement statistics in your clinical trial, send us a mail at statistics@gcp-service.com.