Descriptive statistics summarize a sample, for example the group of patients included in a clinical trial. They have to be distinguished from inductive and explorative statistics, which aim to provide results for interpretation or to support the (main) analysis, respectively. Clinical characteristics, such as the distribution of age or gender, or the proportion of subjects with related co-morbidities can be presented for the sample.
What does summarize mean? In general, the reporting of descriptive statistics of single variables can include scatter parameters, such as the mean, the median, and the mode; and dispersion parameters, which are the range, quartiles, the variance, and the standard deviation. For quantitative data, this usually includes the overall number of patients, and the number and percent of patients within the respective subgroup. For qualitative data the mean, median, standard deviation, quartiles, and extreme values are reported.
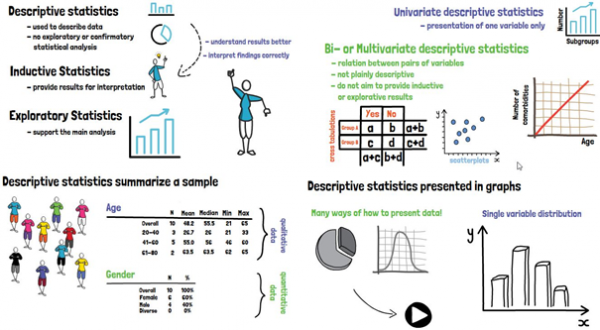
Descriptive statistics can additionally be presented in graphs, of which there are many possibilities, depending on what should or needs to be shown; the most common one being the histogram.
Apart from the above-described univariate descriptive statistics, that present one variable only, there are also bi- or multivariate descriptive statistics, which describe the relation between pairs of variables. These include, amongst others, cross-tabulations, or graphical presentations, for example by scatterplots. These are not plainly descriptive statistics anymore but shall still be mentioned here since they don´t aim to provide inductive or exploratory results.
Descriptive statistics are usually reported for the total sample, for the treatment groups that are being investigated in the study, and for predefined subgroups; for example, by grouping patients according to their country of residence or study site.
If you are interested in a visual presentation of this topic, please feel free to follow us on our GCP Mindset YouTube channel!
If you have any statistical questions, feel free to contact us at: statistics@gcp-service.com


