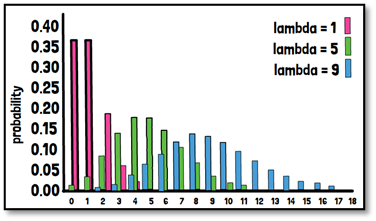
What is the Poisson distribution?
The Poisson distribution is modelling the number of events within a defined time interval or area. For example, counting variables such as the recruitment for
Clinical Research Insights

The Poisson distribution is modelling the number of events within a defined time interval or area. For example, counting variables such as the recruitment for
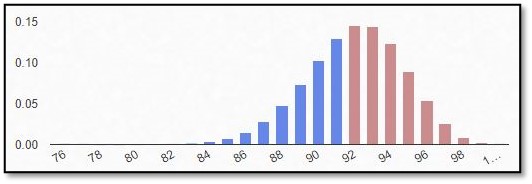
If an outcome variable that can be specified in two response possibilities only, data can be analyzed as binary based on a binomial distribution, where each observation of a defined number of “experiments” represents one of the two outcomes, and the probability of success is the same for each observation.
In this part of our blog series accompanying our #video #series “5 Minutes Statistics for Clinical Trials” we explain what a #binomial #distribution reflects and what are its characteristics.
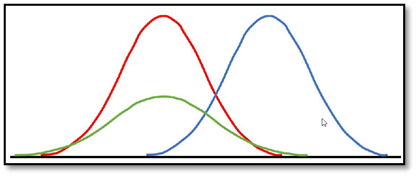
The normal distribution or Gauß distribution is called normal, as it is seen as the ideal case of a distribution. It is showing the distribution
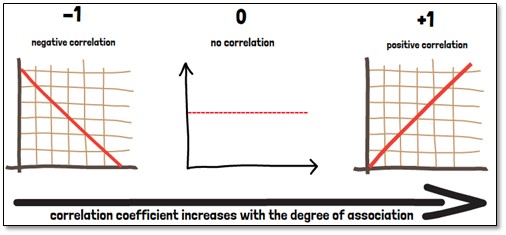
The correlation coefficient (r) is a specific measure that quantifies the strength of a linear statistical relationship between two variables, as well as its direction.
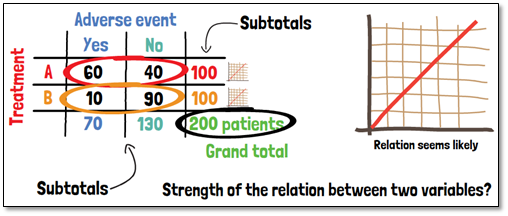
Cross tables are also known as contingency or multidimensional tables. Large amounts of data can be summarized in a structured way and possible relationships between
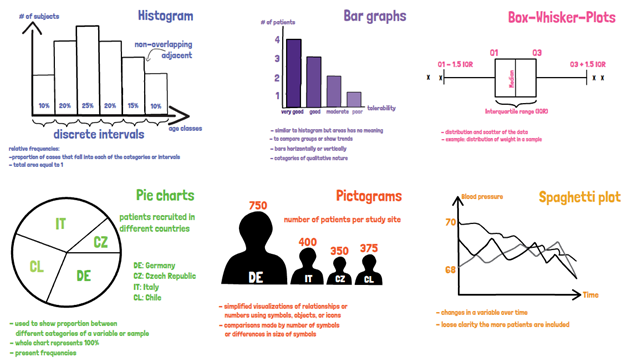
In clinical studies usually a large number of data is generated. The challenging part is then to present the data in a structured and condensed
Descriptive statistics present a good first overview of the data. The frequency of occurrence, as part of descriptive statistics, provides information that is easy to
When presenting the data descriptively, the distribution of single variables can include the following parameters: Scatter parameters such as the mean, median, and mode; dispersion
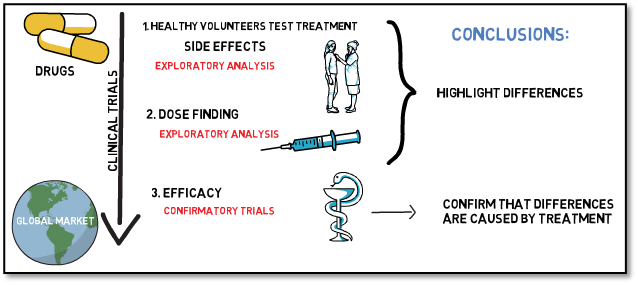
Clinical trials often aim to establish two key parameters about a novel treatment or device: They should show that the product is safe and efficacious.
Descriptive statistics summarize a sample, for example the group of patients included in a clinical trial. They have to be distinguished from inductive and explorative